Sign up for our daily briefing
Make your busy days simpler with the Axios AM and PM newsletters. Catch up on what’s new and why it matters in just 5 minutes.
Catch up on the day’s biggest business stories
Subscribe to the Axios Closer newsletter for insights into the day’s business news and trends and why they matter.
Sign up for Axios Pro Rata
Dive into the world of dealmakers across VC, PE and M&A with Axios Pro Rata. Delivered daily to your inbox by Dan Primack and Kia Kokalitcheva.
Sports news worthy of your time
Binge on the stats and stories that drive the sports world with the Axios Sports newsletter. Sign up for free.
Tech news worthy of your time
Get our smart take on technology from the Valley and D.C. with Axios Login. Sign up for free.
Get the inside stories
Get an insider’s guide to the new White House with Axios Sneak Peek. Sign up for free.
Catch up on coronavirus stories and special reports, curated by Mike Allen everyday
Catch up on coronavirus stories and special reports, curated by Mike Allen everyday
Want a daily digest of the top Denver news?
Get a daily digest of the most important stories affecting your hometown with Axios Denver
Want a daily digest of the top Des Moines news?
Get a daily digest of the most important stories affecting your hometown with the Axios Des Moines newsletter.
Want a daily digest of the top Twin Cities news?
Get a daily digest of the most important stories affecting your hometown with Axios Twin Cities
Want a daily digest of the top Tampa Bay news?
Get a daily digest of the most important stories affecting your hometown with the Axios Tampa Bay newsletter.
Want a daily digest of the top Charlotte news?
Get a daily digest of the most important stories affecting your hometown with Axios Charlotte
Want a daily digest of the top Nashville news?
Get a daily digest of the most important stories affecting your hometown with the Axios Nashville newsletter.
Want a daily digest of the top Columbus news?
Get a daily digest of the most important stories affecting your hometown with the Axios Columbus newsletter.
Want a daily digest of the top Dallas news?
Get a daily digest of the most important stories affecting your hometown with the Axios Dallas newsletter.
Want a daily digest of the top Austin news?
Get a daily digest of the most important stories affecting your hometown with the Axios Austin newsletter.
Want a daily digest of the top Atlanta news?
Get a daily digest of the most important stories affecting your hometown with the Axios Atlanta newsletter.
Want a daily digest of the top Philadelphia news?
Get a daily digest of the most important stories affecting your hometown with the Axios Philadelphia newsletter.
Want a daily digest of the top Chicago news?
Get a daily digest of the most important stories affecting your hometown with the Axios Chicago newsletter.
Sign up for Axios NW Arkansas
Stay up-to-date on the most important and interesting stories affecting NW Arkansas, authored by local reporters
Want a daily digest of the top DC news?
Get a daily digest of the most important stories affecting your hometown with the Axios DC newsletter.
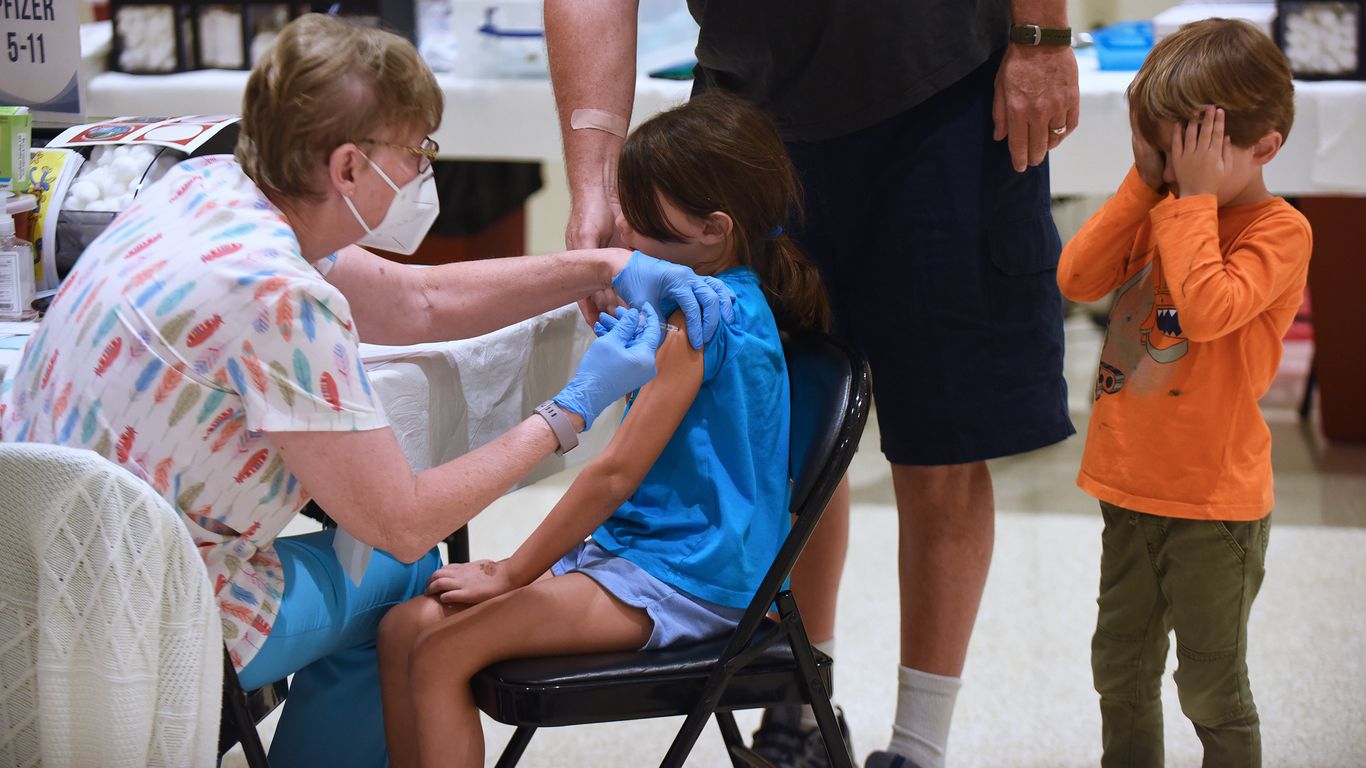
A nurse gives a little girl a shot of the Pfizer COVID-19 vaccine while her brother covers his eyes at a vaccination site in Florida. Photo: Paul Hennessy/SOPA Images/LightRocket via Getty Images
Pfizer and BioNTech on Tuesday asked the Food and Drugs Administration to authorize its COVID-19 vaccine for children 6 months to 5 years old.
The big picture: Hospitalizations of younger children have reached their highest levels since the beginning of the pandemic.
- More than 4 in 100,000 kids under 5 have been hospitalized since mid-December, compared to approximately 1 per 100,000 hospitalizations for children ages 5 to 17.
State of play: Federal regulators could authorize the vaccine and make it available by the end of February, Axios’ Caitlin Owens reports.
- The Pfizer-BioNTech vaccine would be initially available as a two-dose regimen. However, recent data has shown that two shots of the vaccine did not produce a sufficient immune response in children aged 2 to 4.
- An adequate immune response was seen in children 6 months old to 2 years old.
- Pfizer said it would continue to research how well three doses of its vaccine work on younger children.
Sign up for our daily briefingMake your busy days simpler with the Axios AM and PM newsletters. Catch up on what’s new and why it matters in just 5 minutes.Catch up on the day’s biggest business storiesSubscribe to the Axios Closer newsletter for insights into the day’s business news and trends and why they matter.Sign up for Axios Pro RataDive into the world of dealmakers across VC, PE and M&A with Axios Pro Rata. Delivered daily to your inbox by Dan Primack and Kia Kokalitcheva.Sports news worthy of your timeBinge on the stats and stories that drive the sports world with the Axios Sports newsletter. Sign up for free.Tech news worthy of your timeGet our smart take on technology from the Valley and D.C. with Axios Login. Sign up for free.Get the inside storiesGet an insider’s guide to the new White House with Axios Sneak Peek. Sign up for free.Catch up on coronavirus stories and special reports, curated by Mike Allen everydayCatch up on coronavirus stories and special reports, curated by Mike Allen everydayWant a daily digest of the top Denver news?Get a daily digest of the most important stories affecting your hometown with Axios DenverWant a daily digest of the top Des Moines news?Get a daily digest of the most important stories affecting your hometown with the Axios Des Moines newsletter.Want a daily digest of the top Twin Cities news?Get a daily digest of the most important stories affecting your hometown with Axios Twin CitiesWant a daily digest of the top Tampa Bay news?Get a daily digest of the most important stories affecting your hometown with the Axios Tampa Bay newsletter.Want a daily digest of the top Charlotte news?Get a daily digest of the most important stories affecting your hometown with Axios CharlotteWant a daily digest of the top Nashville news?Get a daily digest of the most important stories affecting your hometown with the Axios Nashville newsletter.Want a daily digest of the top Columbus news?Get a daily digest of the most important stories affecting your hometown with the Axios Columbus newsletter.Want a daily digest of the top Dallas news?Get a daily digest of the most important stories affecting your hometown with the Axios Dallas newsletter.Want a daily digest of the top Austin news?Get a daily digest of the most important stories affecting your hometown with the Axios Austin newsletter.Want a daily digest of the top Atlanta news?Get a daily digest of the most important stories affecting your hometown with the Axios Atlanta newsletter.Want a daily digest of the top Philadelphia news?Get a daily digest of the most important stories affecting your hometown with the Axios Philadelphia newsletter.Want a daily digest of the top Chicago news?Get a daily digest of the most important stories affecting your hometown with the Axios Chicago newsletter.Sign up for Axios NW ArkansasStay up-to-date on the most important and interesting stories affecting NW Arkansas, authored by local reportersWant a daily digest of the top DC news?Get a daily digest of the most important stories affecting your hometown with the Axios DC newsletter.A nurse gives a little girl a shot of the Pfizer COVID-19 vaccine while her brother covers his eyes at a vaccination site in Florida. Photo: Paul Hennessy/SOPA Images/LightRocket via Getty ImagesPfizer and BioNTech on Tuesday asked the Food and Drugs Administration to authorize its COVID-19 vaccine for children 6 months to 5 years old.The big picture: Hospitalizations of younger children have reached their highest levels since the beginning of the pandemic. More than 4 in 100,000 kids under 5 have been hospitalized since mid-December, compared to approximately 1 per 100,000 hospitalizations for children ages 5 to 17.State of play: Federal regulators could authorize the vaccine and make it available by the end of February, Axios’ Caitlin Owens reports.The Pfizer-BioNTech vaccine would be initially available as a two-dose regimen. However, recent data has shown that two shots of the vaccine did not produce a sufficient immune response in children aged 2 to 4.An adequate immune response was seen in children 6 months old to 2 years old. Pfizer said it would continue to research how well three doses of its vaccine work on younger children.
